 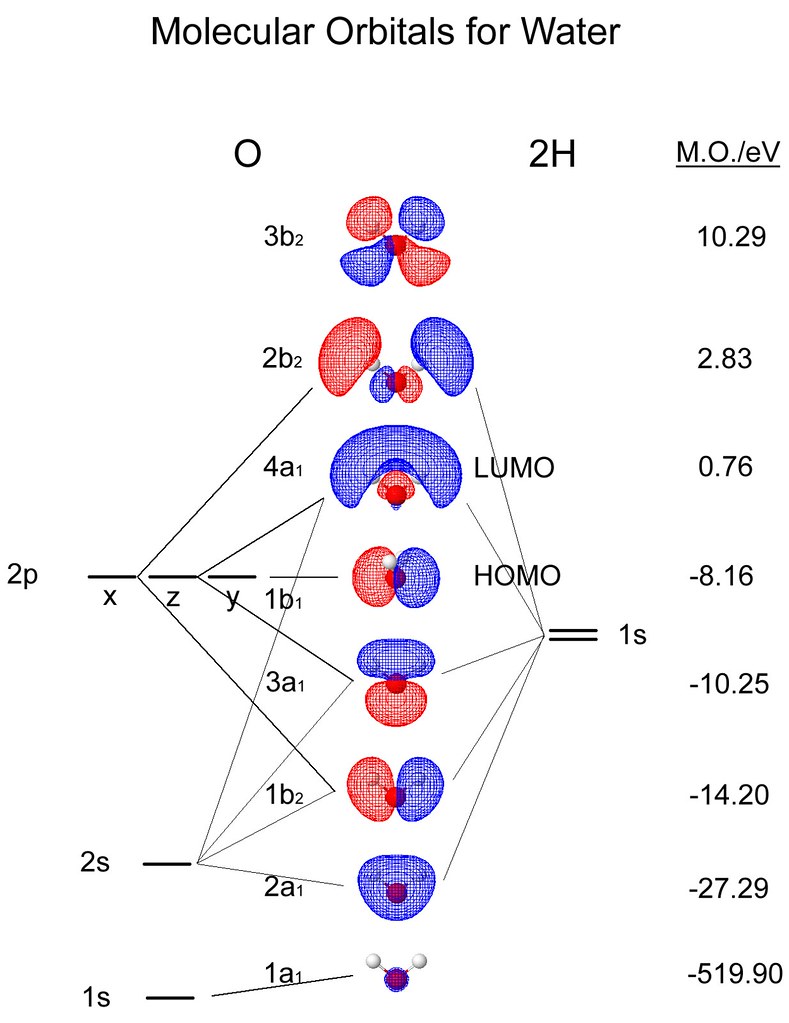
In the helium atom there are two electrons associated with the helium nucleus. The electron has a fixed energy and a fixed spatial distribution called an orbital. With a hydrogen atom the probability distribution is spherical around the nucleus and it is possible to draw a spherical boundary surface, inside which there is a 95% possibility of finding the electron. It is not possible to accurately determine the position of the electron, but it is possible to calculate the probability of findng the electron at any point around the nucleus. Atoms can share one, two or three electrons (forming single, double and triple bonds).Ī hydrogen atom consists of a nucleus (a proton) and an electron. Woodward and Hoffmann work was assimilated into general organic reaction theory.īy sharing electron, molecules can form bonds, and it is possible to regard the sharing of two electrons by two atoms as constituting a chemical bond. 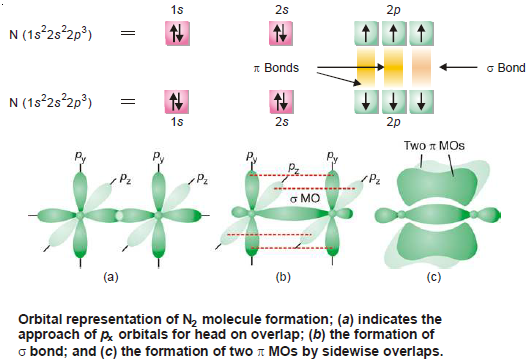
the Highest Occupied, HOMO and Lowest Unoccupied, LUMO) both the regio- and stereospecificity could be accountred for. Woodward and Hoffmann showed that by examining the interaction of the frontier molecular orbitals (ie. Diels-Alder and pericyclic reactions, the curly arrow format is not very clear and in some instances the reactions are described as no-machanism reactions. The use of "canonical forms" and "resonance hybrids", alonng with extensive use of curvy arrows has provided the fundamental background to modern organic theory, but for eg. Pauling with others, developed Resonance Theory, which provided the rationale to an all-embracing orbital theory. Lewis Theory uses curly arrows to denote electron migration during a chemical reaction and has led to a greater understanding of the factors controlling chemical reactions. In order to understand the mechanism of many reactions, Lewis Theory, developed by Robinson and Ingold, can provide a succesful answer. Atoms can be represented as spheres connected by cyclinders or sticks. Modern chemistry has depended upon the use of models of increasing comlexity. The pictorial content uses both Chimed and VRML enhanced images, and demonstrates that these forms can be used in the place of traditional "curly arrows" and "resonance hybrids", as they can provide a deeped and more subtle insight into the mechanism of a reaction. It uses 3-D pictorial presentations of molecular orbitals to elucidate organic reaction mechanisms - such as those found in pericyclic chemistry. This collection of web documents can be used as a "backup" to Henry Rzepa's on-line Pericyclic Chemistry course. Introduction to Molecular Orbital Theory Introduction to Molecular Orbital Theory 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
